LATEST UPDATE: ICAN Confirms “Hot Batches” in Moderna and Pfizer Vaccine Data
ICAN has successfully accessed exclusive, previously undisclosed batch and dosage information for the Moderna and Pfizer COVID-19 vaccines. This information enables the identification of the quantity of doses distributed for each batch number. OpenVAERS, analyzing the data acquired by ICAN, has identified numerous “high-risk batches.”
Since March 2022, ICAN has been pursuing complete disclosure of the batch and dose details for the Moderna and Pfizer COVID-19 vaccines. Previously, the absence of this data hindered the ability to pinpoint “high-risk batches” through VAERS adverse reaction reports, as the distribution quantity for each batch was unknown. Now, with this newly obtained data, it’s possible to calculate the incidence of reported harm per batch.
ICAN shared that its legal team has successfully obtained the necessary data from the CDC to ascertain the distribution count of doses per batch.
The journey to acquire this information spanned over two and a half years, involving numerous FOIA requests and a lawsuit, which led to the initial release of some data in June 2022, October 2022, and July 2023. However, ICAN, dissatisfied with the CDC’s failure to provide key data, pressed on with appeals for both Pfizer and Moderna batch data. Given ICAN’s history of legal action, the CDC likely anticipated a lawsuit if the appeals were rejected. Consequently, on December 13, 2023, the CDC relented and unveiled the updated data sets.
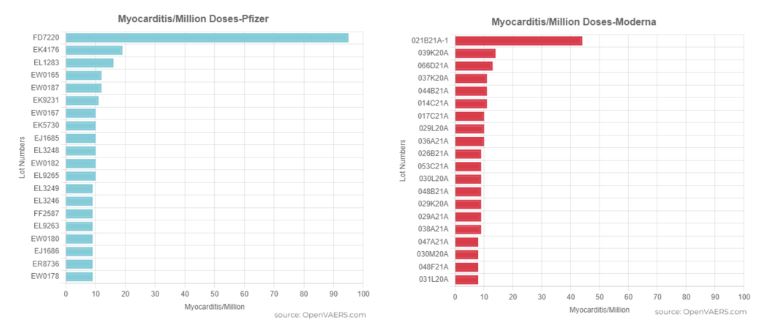
ICAN then shared this information with OpenVAERS, who, upon correlating it with VAERS data, conducted analyses that suggest certain batches may be linked to a disproportionately high number of adverse reactions. For instance, their graph indicates that specific batches from both Pfizer and Moderna were associated with a notably high incidence of myocarditis reports.
The Moderna and Pfizer data are available for download, and OpenVAERS’ comprehensive analysis of the data can be reviewed. ICAN urges all interested entities to perform their analyses using the raw data. Updates regarding J&J batch data and analyses will be forthcoming.





From January 10, 2024 in the Dailymail.
https://www.dailymail.co.uk/news/article-12942189/Covid-vaccine-effects-Facebook-censoring.html
”The Covid vaccine gave me side effects that ruined my life, but Facebook keeps censoring me from telling my friends what happened”