Letter to Wyoming Attorney General: Consider State Criminal Investigation of CDC Officials for Reckless Endangering
Attorney Edward A. Berkovich wrote a November 26, 2022, letter to Wyoming Attorney General Bridget Hill stating that there may be reasonable suspicion to investigate Centers for Disease Control and Prevention (CDC) officials for recklessly endangering Wyoming residents by waiting three months to alert the public to the risk of …
Report 46: How Many Pregnant Women Received LNP/mRNA via COVID-19 Vaccine During the Year 2021? Only Estimates Are Available.
Shockingly, as 2022 comes to a close and the world reaches the two-year mark of experimental COVID-19 vaccines being available to the general public, the scientific and medical communities, as well as everyday citizens, still have very little information about the effects of mRNA COVID-19 vaccines on pregnant women and …
Letter to Florida Attorney General: Consider State Criminal Prosecution of CDC Officials for Reckless Endangerment of Citizens
Attorney Edward A. Berkovich wrote a November 17, 2022, letter to Florida Attorney General, Ashley B. Moody, copying State Surgeon General, Joseph Ladapo, MD, asking her to consider screening for prosecution of the Centers for Disease Control and Prevention (CDC) officials for violating Florida’s culpable negligence criminal code section. ag …
189 “Fringe Scientists” Fight for the European Parliament to Recognize the Rights of Children During the COVID-19 Pandemic
189 is definitely not a perfect number. It could have been 100 – as in the “100 days to deliver a new vaccine”, or two – as in “two weeks to flatten the curve” or three – as in “three months to save the NHS”, or other such winning number. …
A Drug Safety Physician’s Correspondence with the FDA: A Quest for Accuracy in Investigational Bivalent mRNA COVID-19 ‘Vaccine’ Adverse Event Reporting
Introduction On November 4, 2022, the Centers for Disease Control and Prevention (CDC) and U.S. Food and Drug Administration (FDA) concluded that, based on Vaccine Adverse Events Reporting System (VAERS) and v-safe data received between August 31, 2022, and October 23, 2022, “Health care providers and patients can be …
Letter to the FDA: Vaccine-Induced Immune Thrombotic Thrombocytopenia (VITT), a New and Dangerous Syndrome from Certain COVID Vaccines – Do We Have Them All?
Introduction WHAT IS “VITT”? Vaccine-induced Immune Thrombotic Thrombocytopenia (VITT) is “a newly described syndrome” “characterized by new onset thrombocytopenia [i.e., low blood platelet count] and acute venous or arterial thrombosis [i.e., blood clot], including at unusual sites such as the cerebral venous sinus, with onset of symptoms approximately …
Who Has Been Teaching Humans to Make Terrible Decisions Related to COVID-19?
Photo by Vladislav Babienko on Unsplash.com. Who Has Been Teaching Humans to Make Terrible Decisions Related to COVID-19? Much like the London blitz in World War II, we are getting bombarded. We are under constant bombardment, and yet we do not know it. Just today I was driving and, …
Report 44: Is mRNA-LNP Vaccine-Induced Immunity Inheritable? A Preprint Study Shows It Is.
Summary Some traits acquired via the mRNA-LNP injections are passed genetically from parents to their offspring. The implications of this new finding are profound. Because of this inheritability, mRNA gene therapies – including mRNA “vaccines” – must be prohibited, at least until more is known, for expecting mothers as well …
Reading, Understanding, and Interpreting Medical and Scientific Literature – Assisting the Neophyte.
Recently Modern Discontent published a paper on Substack that proves to be a worthwhile guide for those new to reading medical and scientific literature. “Modern” represents himself/herself as a: “Disaffected Scientist, Disaffected Liberal. Trying to make sense of things and ranting about science, culture, and the world beyond.” https://moderndiscontent.substack.com/p/how-i-tackle-reading-papers …
Impotence, Erectile Dysfunction, and Multiple Prostate Problems Appear as Adverse Events After Vaccination with Pfizer’s mRNA COVID Vaccine
Shocking harms to men’s reproduction and sexual function continue to surface. In this video, DailyClout’s COO and Program Director of the War Room/DailyClout Pfizer Documents Analysis Project, Amy Kelly, talks about prostate problems, erectile dysfunction, and functional impotence found in an untitled Pfizer adverse events document (filename: “125742_S1_M5_5351_c4591001-interim-mth6-adverse-events”). The document is …
Documentary: “Safe and Effective: A Second Opinion”
“Safe and Effective: A Second Opinion” shines a light on Covid-19 vaccine injuries and bereavements, but also takes an encompassing look at the systemic failings that appear to have enabled them. We look at leading analysis of pharmaceutical trials, the role of the Medicines and Healthcare Regulatory Agency (MHRA) in …
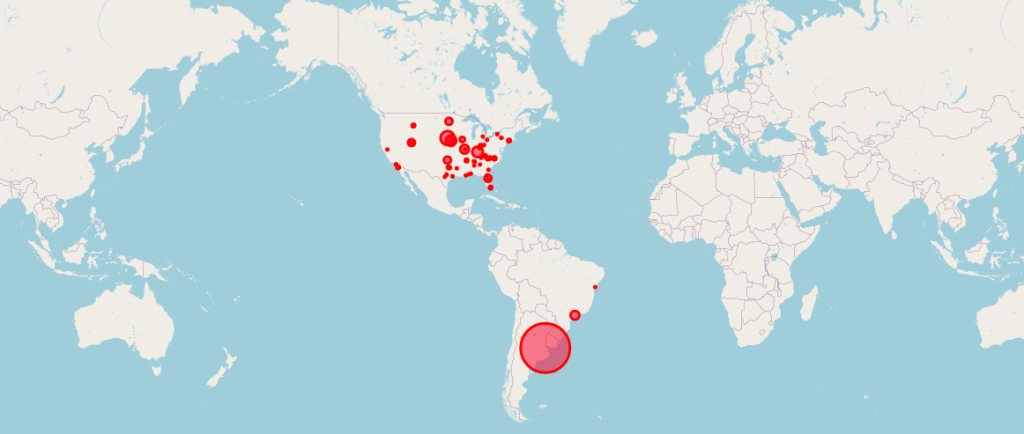
Report 42: Pfizer’s EUA Granted Based on Fewer Than 0.4% of Clinical Trial Participants. FDA Ignored Disqualifying Protocol Deviations to Grant EUA.
Locations of the 170 Pfizer Clinical Trial Patients Used to Gain Emergency Use Authorization (EUA) for Its COVID mRNA Vaccine. Courtesy of OPENVAET.org For audio-visual explanations of this report, please see these videos. So much has been written about the pivotal Pfizer Trial for COVID-19 (C4591001), that it is sometimes …
Report 41: 2021 CDC and FDA Misinformation – Retroactive Editing, Erroneous Spontaneous Abortion Rate Calculation, Obfuscation in the New England Journal of Medicine
The following article is a follow-up to Dr. Chandler’s report, “Data Do Not Support Safety of mRNA COVID Vaccination for Pregnant Women,” which reviewed “Preliminary Findings of mRNA Covid-19 Vaccine Safety in Pregnant Persons,” New England Journal of Medicine, April 21, 2021, and June 17, 2021. To best understand this …
The Sentient World Simulation – Was It Used for the COVID-19 Pandemic Response?
A world simulation was developed for the US military. If this technology was not used to find the optimum Covid pandemic response, then it was a missed opportunity. But, if the technology was used, there are sinister implications. In 2007, Frank Catanzaro, the Chair of Cyber Futures, presented his “Mirror …
Report 40: Data Do Not Support Safety of mRNA COVID Vaccination for Pregnant Women
Commentary on “Preliminary Findings of mRNA Covid Vaccine Safety in Pregnant Persons” as Reported by the Centers for Disease Control and Prevention and the Food and Drug Administration, June 17, 2021, New England Journal of Medicine Note: On September 26, 2022, Dr. Chandler published a follow-up report to the report …
Report 39: Despite Incomplete Safety Trials, the Food and Drug Administration (FDA) Grants Full Approval to Pfizer-BioNTech’s COMIRNATY® for Adolescents 12-15 Years of Age
Without a completed safety study or expert committee review, the FDA issued a supplemental Biologics License Application (“sBLA”) approval letter granting full FDA approval to Pfizer-BioNTech’s COMIRNATY® COVID-19 mRNA vaccine for use in children ages 12-15. This was done even though safety study completion, on which approval should be based, …
Report 43: Twenty-Two Cases of Rare Myocarditis by February 2021, Yet Pfizer Said No “New Safety Issues.” FDA Waits Until June 25, 2021, to Include Myocarditis Risk in Fact Sheets.
As initially reported by Chris Flowers, M.D., on DailyClout.io in April 2022, myocarditis – inflammation of the heart muscle (a.k.a., myocardium) that can reduce the heart’s ability to pump blood as well as cause chest pain, shortness of breath, and rapid or irregular heart rhythms (a.k.a., arrhythmias) [https://www.mayoclinic.org/diseases-conditions/myocarditis/symptoms-causes/syc-20352539] – …
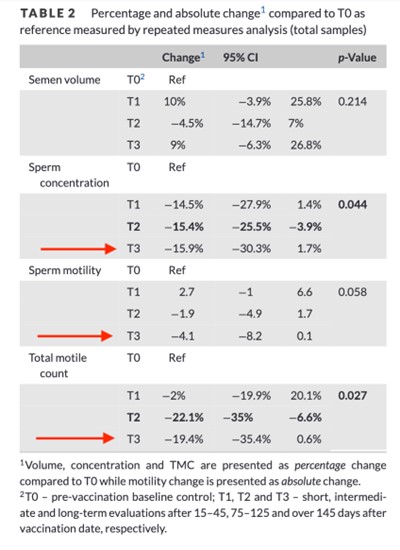
Report 37: Pfizer, FDA, CDC Hid Proven Harms to Male Sperm Quality, Testes Function, from mRNA Vaccine Ingredients
When the COVID-19 vaccine rollout to the public began in late 2020, medical professionals, public health agencies, and government spokespeople all assured the American public that the novel mRNA vaccines did not cause negative systematic effects to human bodies. They promised the public, many of whom were skeptical about the …
Report 03: Phase 1 /2 Study of COVID-19 RNA Vaccine BNT162b1 in Adults: Key Processes Missing.
Mulligan, Lyke et al. Nature Published online August 12, 2020. Cite this article as: Mulligan, M. J. et al. Phase 1/2 study of COVID-19 RNA vaccine BNT162b1 in adults. Nature https://doi.org/10.1038/s41586-020-2639-4 (2020). P1 p2: The authors’ put forth the argument that mRNA in BNT162b1 (Note this series used BNT162b1 …