A Question and Answer Document on VAERS as a Pharmacovigilance Tool
Dr. Jessica Rose – A short Q and A – August 9, 2022
1. “Are the COVID vaccines causing injuries, what kinds, and how do we know?”
Yes. Any human physiological system that you can name, able to be affected in an injurious way, is being affected in an injurious way in a percentage of individuals. We know because VAERS – a pharmacovigilance tool – currently has an extraordinarily high number and range of adverse event reports in the context of the COVID-19 injectable products. We also know because the Yellowcard and EUDRA adverse event (AE) data collection systems are reporting the same things.[1]
2. An introduction to VAERS: what is it, who runs it, what is its history?
VAERS[2] is a pharmacovigilance tool designed to detect safety signals (not detected in pre-market testing) emitted in people following administration of biological or pharmaceutical products. It was originally put into place in 1990 as a solution to non-liability of pharmaceutical companies as per the National Childhood Vaccine Injury Act of 1986 (NCVIA)[3]. In spite of the fact that the NCVIA requires health care providers and vaccine manufacturers to report to the U.S. Department of Health & Human Services (DHHS)[4] specific AEs following the administration of vaccines outlined in the Act, under-reporting is a known imperfection of the VAERS system.[5],[6] VAERS is maintained and monitored by the Centers for Disease Control (CDC)[7] and the Food and Drug Administration (FDA)[8].
3. In fairness to the “other side”, what is the distinction between reported injury and confirmed injury?
A reported injury that gets into the front-end downloadable database IS a confirmed injury.[9]
4. What is the reality of submitting an adverse event report to the VAERS system (the difficulty and the time investment involved on front end and in follow up)?
It takes 30 minutes – at least – to file a VAERS report online. It requires a multi-electronic page submission of detailed information and each electronic page is time-restricted. This means that if one does not complete the electronic form fast enough, one must begin the submission process again. The vetting process is long and dealt with by a handful of contracted employees.[10] There is a massive backlog of data in VAERS.[11]
5. What percentage of actual incidents is reflected by the number of reports – and how are we making that guess?
The under-reporting factor represents the percentage of individuals who actually experienced an adverse event in contrast with how many individuals reported an event. Studies have shown that the percentage of incidents reported can be quite low (1–10%). My own study showed an under-reporting factor of 31, which means that each stand-alone adverse event count must be multiplied by a factor of 31.[12],[13],[14] Steve Kirsch also estimated the under-reporting factor to be 41 based on Anaphylaxis data.1
6. What are the top injuries reported with percentages?
Cardiovascular and neurological injuries remain highest with regard to grouped adverse event reporting frequency.[15],[16]
7. How does this injury rate compare to all other vaccines recorded by VAERS?
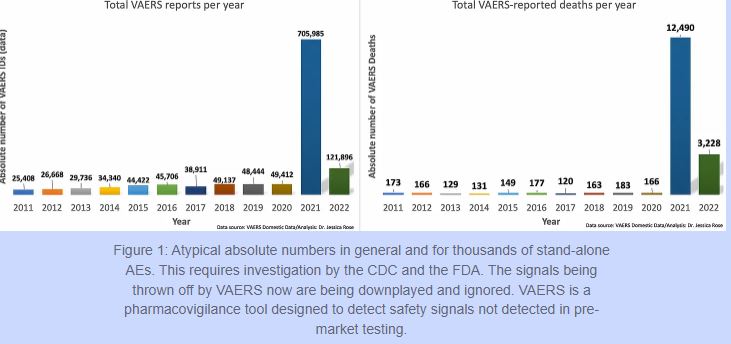
The injury rates do not compare in any way, shape or form to all other vaccines combined for the past 30 years. For the past 30 years, the VAERS data has housed approximately 39,000 adverse event reports per year for all vaccines combined. Of these, approximately 155 have been deaths. In 2021, there were 705,985 reports and 12,490 deaths in the context of the Moderna[17], Pfizer/BioNTech[18] and Janssen[19] products, in the VAERS Domestic data set alone, not including all other products on the vaccine list.




I am able to create $88/h to complete few jobs on home computer.e3 I’ve never thought that it’s even achievable but my closest mate earning $25k only within five weeks simply working this leading project & she had convinced me to join…Discover extra details by going following link http://Www.NetJob1.com